|
At the Brigham and Women’s Hospital we met Dr. Mizuno who is a co-founder of Histogenics with a PhD, working in orthopedic research at Harvard Medical School. Dr. Mizuno explained his involvement in the research he has done that led to the development the product NeoCart;
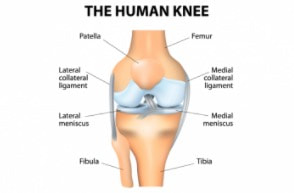
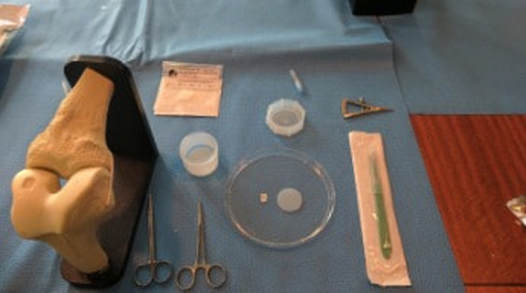
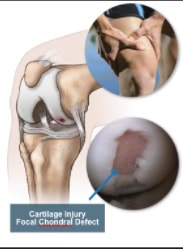
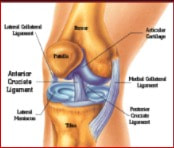
The idea he had was to take patients cells and biomaterials, and then put them into a bioreactor. The bioreactor is used to replicate the pressure of walking in order to help grow the knee cartilage. Dr. Mizuno explained the development of the machine that he created with other Japanese inventors and makers of the bioreactor; Today Dr. Mizuno continues his research at Harvard, in order to further develop regenerative medicine products for the future Developing a product in a new area of science is not easy. It requires that your ideas work, but also that your company can get the money, research and clearance from the government together as well. Doing clinical trials, for example, takes a long time, and a lot of money - without the backing from the financial sector, the project would never get off the ground. At Histogenics, the financial department at Histogenics communicates with each other branch of the company to create budget plans, as well as contracting and attracting investors, all to make sure the company can continue running. Financial employees learn about what the other departments do and what they need, then use the information to plot out the company’s purchases and operations, accommodate for new employees or facilities, and communicate with investors to ensure the company has the money it needs to meet these demands. They stay informed on the market situation and competitors to Histogenics as well. The Clinical Trial process involves the physical cartilage implantation surgery as well as the preparation for post op physical therapy treatment for the patient. The initial step of the surgery is to determine the area of the cartilage that is unhealthy, and remove its remains. The defect is in turn surrounded by healthy cartilage. The Neocart is then cut according to size and shape of the patient’s defect. To improve efficiency, surgeons first use foil to create a template of the affected area. To operate on the defect, it may be necessary to bend the patient’s knee at a specific angle. The defected area is covered in the CT3 bio adhesive on both top and bottom of the Neocart before using the curing component to solidify the healthy cartilage.. Following the surgery, the patient will spend six weeks in rehabilitation, which is much quicker and easier than the existing cartilage recovery process. I learned that a “quick and easy” surgery is extremely hard.
The facilities at the Lexington location manufacture collagen for use in both the scaffolds and the CT3 adhesive for the NeoCart product. In accordance with cGMPs (current Good Manufacturing Procedures), this site utilizes an ISO Class 8 clean room with proper gowning routine and a USP water purifier in order to produce a high quality product. For scaffolds, once the mixture is made it is freeze dried until it resembles something similar to styrofoam. This is then sliced to a 1.5 mm sample from which the NeoCart scaffold and several surrogates can be harvested. For the CT3 adhesive, methylated collagen is mixed with PEG (premade) in order to create a biodegradable substance that will hold the NeoCart in place until the cartilage can be held in on its own. At the left, we're getting on our gowns - even though this site is cleaner than a surgical room in a hospital, it's still more relaxed than the lab in Waltham. Juliana and Hannah like science!
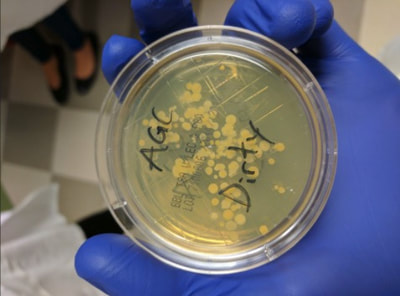
Today, we got to work with some of the equipment that the scientists at Histogenics actually use in their work. In our lab activity, we prepared the collagen solution that would become the gel that sets the cells into the NeoCart scaffold. In doing this activity we learned how to use equipment such as micropipettes, vortexers, and sterile gloves. Afterwards, we used agar plates to sample microbes on our hands compared to the sterile gloves, which we will review later this week. We had a look at a second lab that houses the bioreactors which stimulate the NeoCarts by simulating the stress and pressure from walking on a knee. At the left, all of us are using pipettes to precisely measure out liquid samples. In the middle, Mr. Kennedy is explaining how they company makes sure that its quality control is as perfect as possible. At the right, the bacteria on Andrew's hands has gone crazy - it's pretty clear why everyone takes being clean so seriously!
Today, we learned how to put on sterile gloves. Don't laugh - the entire operation of growing cloned tissue would be ruined if people weren't super careful about making sure everything was incredibly clean.
The manufacturing process is a precise procedure that ensures that the NeoCarts are sterile and ready for implantation. The process takes place in what is called the clean room. In the clean room, several steps and precautions are followed to ensure cleanliness and prevent any risk of contamination. Every surface is sprayed nightly to kill bacteria, and HEPA air filters work constantly to remove 99.99% of particles .5 microns or larger, adhering to regulations for ISO Class 7 clean rooms. Anyone entering must gown up head to toe and at first must pass certification to show that the gowning process is carried out correctly, including clean up crew who come in after hours. Regenerative medicine is a revolutionary branch of tissue engineering and molecular biology that aims to use the body’s own resources along with additional outside products to treat diseases or injuries. Histogenics is a company that is seeking to use regenerative medicine to specifically treat cartilage related injuries in the knee. NeoCart, Histogenics’ main product, is a sample of new cartilage regrown using the patient’s own cells. The NeoCart is grown in a bioreactor that replicates the physical environment of the knee so it can be safely and effectively produced and then implanted. Today we met everyone in the group, and met the rest of the students. Adam Gridley gave us an introduction to the company, and explained the field of regenerative medicine to us. We also toured the Histogenics location, and saw the incubators and the hydrostatic pressure bioreactor.
|
Andrew Carr
|









 RSS Feed
RSS Feed